
At 7 months old, Remy Miller was diagnosed with spinal muscular atrophy.
A new drug and a team at Children’s Hospital of Richmond at VCU changed her life.
Remy Miller sits up, kicks her feet and holds her bottle like other 20-month-old toddlers would at this stage of development. Four months ago these types of milestones weren’t possible for Remy, but a lifesaving infusion of the drug Zolgensma at Children’s Hospital of Richmond at VCU changed her life forever.
Remy suffers from the most severe form of spinal muscular atrophy, a rare genetic disease characterized by severe muscle weakness, trouble breathing and swallowing, and failure to achieve important milestones such as sitting without support, eating, speaking, crawling and walking. SMA is a leading genetic cause of infant death.
Zolgensma is the first gene therapy approved to treat children younger than 2 with SMA. The product acts on the gene Remy is missing by using an adeno-associated virus that has been modified to infect cells to deliver the gene she is missing so she can make her own protein. The therapy received Food and Drug Administration approval on May 24. CHoR is the first hospital in central Virginia to administer Zolgensma.
Remy’s doctor, Amy Harper, MD, director of the Pediatric Neuromuscular Clinic at CHoR, knew the drug’s manufacturer, AveXis, was running several ongoing studies. “Other patients I had been able to get into the trials to get the medicine quickly had such good results,” Harper said. Unfortunately, the AveXis study that Remy was eligible for was no longer taking patients.
Harper made the SMA diagnosis when Remy was 7 months old.
Her parents noticed she didn’t have good neck control and wasn’t sitting up like other babies. “We thought it was because she was premature,” said Nicole Arani, who delivered Remy at 35 weeks.
Arani and Remy’s father, Brett Miller, were referred to Harper. Hearing Remy’s diagnosis was devastating for her parents. “We were in shock and denial,” Miller said. “That was the first time in my life where I felt I had zero control of everything from my life to my daughter’s life. I knew everything would be different. It would be an uphill fight.”
Harper knew that time was of the essence in treating Remy. “Progression of the disease can result in respiratory failure. A common cold can precipitate this at any time,” she said.
If untreated, babies with the most severe form of SMA will die by age of 2 unless they are on a ventilator and have a feeding tube. Delivering gene therapy to save dying motor neurons is time sensitive. “That’s why it’s crucial to intervene with therapies at the earliest possible time following diagnosis,” Harper said.
When Harper couldn’t get Remy into a clinical trial she decided to use the drug Spinraza while waiting for FDA approval of Zolgensma.
An infusion of Spinraza was administered to Remy’s back every two weeks at first. “Then it was monthly and then every four months,” Arani said. “It would be like an all-day visit in outpatient.”
Remy responded to the treatment but was still not holding up her head. She could sit propped up for only short periods of time. “The drug slowed down the progression of losing muscle while we waited for treatment,” Arani said.
After Harper met with the family to discuss the risks and benefits of Zolgensma — the main side effect is liver toxicity — Remy’s parents decided it was the best option.
“We all agreed this was the next step that we wanted to take and began the process of insurance approval,” Harper said. “It was an extensive authorization process with multiple levels of appeals, numerous phone calls and several peer-to-peer phone conversations.”
Coordinating the one-time infusion was a team effort that began with a multiteam meeting “that included about 20 people at the table,” Harper said. “We had to have a system that would work for the infusion and we were ready to go when the medicine arrived.”
The drug had to be sent to CHoR in a special transport box. It has to be prepared by weight in order to have an accurate dosing and infused in a special setting to minimize patient risk. “It took a village to get the process in place,” Harper said.
Donna Palmer, clinical coordinator of the infusion center at CHoR, had to consider multiple factors in the planning. “I looked at staffing and who would be most suited to administer the infusion,” she said. “We had to plan a day where the only negative pressure room in the Children’s Pavilion would be available. We knew the infusion had to be on a Tuesday or Thursday when the room was not being used by the dialysis department.”
Palmer and her team had heard that Remy’s veins would be challenging and Palmer didn’t want to worry about getting the two protocol-mandated IVs started on her own.
“That’s not typical of what we do,” she said of the two IVs. “I reached out to the Pediatric Emergency Department nurses and pediatric anesthesia to see if they could help with the IV start and I could focus my attention on the infusion. I’m glad I did that because the emergency department nurses also found her veins difficult to access and anesthesia was needed to place the second IV.”
With the two IVs in place, Palmer got ready to deliver the one-hour infusion. “I didn’t want to let myself think of this as a lifesaving infusion. I wanted it to be as normal as possible. I didn’t want to get anxious and worked up about it,” she said. “I kept telling myself, ‘this is what you do every day.’”
But it was an emotional time for Palmer, who counted down the minutes of the infusion.
“It was so monumental,” she said. “When I finished, I had a sigh of relief that it went smoothly.”
Remy had to be observed for six hours after the infusion to ensure that her vital signs were stable and she wasn’t having any problems. “That’s a long time for a toddler to be in a room,” Palmer said. “We had a child-life therapist come in and she set up a play mat and handheld toys Remy could play with. She also got her favorite videos. Remy was having a ball playing and eating her snacks.”
This was the first time Palmer had given an infusion of this magnitude. She was honored to be part of the history-making process. “It’s such a privilege to do something that is so life changing,” she said. “I was touched by it and thrilled to do it.”
The infusion was given top priority. Even Harper worked from the infusion room that day.
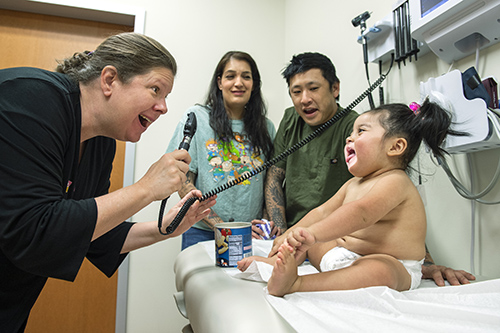 After the infusion, the risk for liver toxicity is high so Harper had to see Remy on a weekly basis. “Her family and I have been watching her closely,” Harper said. “What I am seeing is she is not losing any abilities and seems to be improving. She can sit more upright with a straight back and maintain neck and head control. She can reach over her head to get her snacks, a big motivator for her. This drug is not a cure but the improvements in the quality of her life are priceless. She will now be able to live a longer and fuller life.”
After the infusion, the risk for liver toxicity is high so Harper had to see Remy on a weekly basis. “Her family and I have been watching her closely,” Harper said. “What I am seeing is she is not losing any abilities and seems to be improving. She can sit more upright with a straight back and maintain neck and head control. She can reach over her head to get her snacks, a big motivator for her. This drug is not a cure but the improvements in the quality of her life are priceless. She will now be able to live a longer and fuller life.”
When Remy visits, she can be heard throughout the clinic. “She likes to tell everybody goodbye. She says that from start to finish,” Harper said.
Remy’s illness has brought the family closer together. Miller said it helped him appreciate the smaller things in life. He and Arani are hopeful the drug will prolong Remy’s life expectancy. “The infusion changed her course of life,” Miller said. “From here on out, it’s only going upward. She gets stronger every day.”
They now see a sparkle in their daughter’s eyes because everything “is new to her,” Miller said.
“I think this whole time she had an idea of what to do but hasn’t been able to physically do it,” he said. “We wouldn’t have gotten the drug if not for Dr. Harper and her team. They put forth 110 percent effort. We couldn’t ask for a better team of doctors. They were there for us. They put in their all to work for kids and families.”
